Featured Blogs
On April 9 and 10, the Medicare Payment Advisory Commission (MedPAC) met for their April meeting, the last meeting in the 2026 Cycle. Commissioners discussed several topics including, how to improve payment incentives, the impact of Medicare Advantage (MA) on hospitals’ and post-acute care (PAC) providers’ finances, and institutional special needs plans (I-SNPs). Commissioners all expressed concerns with the current system and voiced numerous solutions.
IMPROVING PAYMENT INCENTIVES IN MEDICARE
MedPAC staff began this session discussing the drivers of Medicare’s spending growth, payment incentives within fee-for-service (FFS), alternative payment models (APMs) and MA, as well as recommendations to improve Medicare’s incentives. These recommendations can be viewed on slides 29-38 in the Improving Payment Incentives” slideshow found here. MedPAC staff shared that its presentation, along with the Commissioners’ discussion, will be included in the MedPAC June 2026 report to Congress.
Overall, Commissioners agreed on the shortcomings of FFS, APMs, and MA, saying that they each have their strengths and weaknesses when it comes to payment incentives. Commissioners expressed that FFS is foundational, in that it serves as a guardrail to ensure MA provides a variety of services, yet it leads to high inflation. Commissioners shared that APMs are an incentive layered on top of FFS and have tools to avoid unnecessary plan expenses but do not have sufficient funding to support the infrastructure needed. In terms of MA, a few Commissioners said that it is effective at controlling costs, but the incentive is currently centered around coding care rather than quality of care. Commissioners concluded that each of these services play a core role and are vital but agreed that there needs to be stronger tools to provide better incentives to providers.
Additionally, Commissioners emphasized the need to ensure beneficiaries know they are being enrolled in Medicare, MA, or APMs so they can best understand the benefits available to them. There was also concern regarding the rise of costs as people age and are diagnosed with additional chronic illnesses. Commissioners recommended payment mechanisms that encourage specialist-led services with more focus on patients and increased specialist engagement with APMs to combat the rising costs. A few Commissioners mentioned rising drug costs and expressed concern over the focus on price and quantity impacts, rather than innovation impacts. A few Commissioners noted that there are tools in place to manage every cost except for drug costs and recommended the utilization component as an incentive to control when drugs are used in a managed environment.
MA ENROLLMENT AND HOSPITALS’ AND POST-ACUTE CARE (PAC) PROVIDERS’ FINANCES
MedPAC staff began this session giving a brief background on MA enrollment and plan incentives, then discussed how MA enrollment affects hospital finances and PAC providers’ finances. The staff explained the elements of the study they conducted as there was no pre-existing literature that included all of the information they needed. Their research showed that there is no significant association between MA penetration and hospitals’ and PAC providers’ finances, despite complaints that providers are experiencing difficulties navigating the space. This chapter will be included in the June 2026 Report to Congress.
A number of Commissioners voiced concerns on the subjectivity of what is medically necessary, specifically in terms of referrals and denials. One Commissioner detailed that providers have different resources they use to evaluate medical necessity than health plans do, which contributes to different decisions. One Commissioner explained that an MA plan can decide a procedure is not necessary if it’s nonemergent, which leads to increased time-to-placement, which increases costs. While the overturn rate of these denials is very high, providers then lose money and time appealing the decision. Other Commissioners echoed concerns about this. One Commissioner questioned if the reason data is not reflecting the issue is due to the way providers and hospitals have adjusted to MA market penetration in order to combat the rising costs.
Commissioners also spent time discussing heterogeneity between MA plans, and between home health agencies (HHAs), skilled nursing facilities (SNFs), and inpatient rehabilitation facilities (IRFs). Commissioners explored how MA plans vary in how they approach situations depending on the geography and relationships with providers in the areas they are operating in. Commissioners noted that each facility varies in size, coverage, and care level and that these differences impact the solutions they need. For example, one commissioner detailed that smaller SNFs operate better than larger SNFs, while smaller HHAs are not as effective as larger HHAs. Commissioners continuously raised this concern and there was agreement about the need to look into the structural failures currently in place that are either positively or negatively impacting each player.
INSTITUTIONAL SPECIAL NEEDS PLANS (I-SNPS): PROVISION OF SERVICES, NETWORK-ADEQUACY REQUIREMENTS, AND STAR RATINGS
MedPAC staff began this session reviewing their work last year on I-SNPs. Staff also emphasized that this work will not be included in the June 2026 Report to Congress, but if there were commissioner interest, they would conduct additional work in the next cycle and include this as a chapter in the June 2027 Report.
A number of Commissioners voiced concerns about the interaction between Medicare and Medicaid plans within I-SNPs as it is the only model that can apply to long-stay older adults on Medicaid that are getting care in nursing homes from Medicare. One Commissioner specifically raised the issue of conflicting incentives in terms of nursing homes electing to send patients to the hospital due to higher reimbursement rates from Medicare versus I-SNPs focus on keeping care within the nursing home. Additionally, Commissioners expressed the need to better understand what nurse practitioners are doing and how they interact with the rest of the nursing home staff.
Other Commissioners also asked questions regarding the functionality of I-SNPs and the data collected on them. One Commissioner asked if there was a way to identify incident to billing in nurse practitioner visits, to which the staff explained that they are salaried employees, so it is unclear if they are cataloging every visit. Another Commissioner asked why insurer sponsored I-SNPs are getting higher star ratings over those that are provider sponsored. Staff explained that insurer sponsored I-SNPs are simply larger, so they have more enrollees and therefore get more data. Additionally, one Commissioner proposed having CMS require SNP beneficiaries, or their proxy, be educated about the choice of FFS, MA, or I-SNP coverage within the nursing home.
Today’s sessions concluded with a public comment period. Shannan Wu, the Director of Payment Policy at the American Hospital Association urged MedPAC to continue carefully examining the role MA plays in access to care and spending. Wu explained that the major high-cost drivers in Medicare can be found in MA, despite the greater administrative burdens, and believes the Commission should look towards combatting that.

What do President Barack Obama, former Virginia Governor Glenn Youngkin, and former military personnel and Veterans have in common?
No, that’s not the punchline for a bar joke in the DC metro area – but it is about a brawl going on in the DMV. It’s about redistricting.
What’s Going on in Virginia?
Virginians are currently voting on whether to amend their state constitution to allow for mid-decade redistricting. If the amendment passes, the General Assembly will be granted the power to conduct mid-decade redistricting through 2030, a process normally handled by an independent commission. The effect would be a new Congressional election map more favorable to Democratic candidates for November.
Remind Me Again about Redistricting
Redistricting is the process of redrawing congressional district boundaries, which typically occurs every 10 years after the decennial census. However, there are exceptions to this. Courts may order a state to redistrict on the grounds that a map violates state or federal law. States have also, on rare occasions, engaged in mid-decade redistricting, if allowed, for partisan gain.
Don’t Mess with Texas
The current mid-decade redistricting fight began in the summer of 2025 when the Texas legislature instated a new map to create additional GOP seats. In response, California held a special election to approve a new map to create additional Democratic seats. Many states have gotten involved since. As of March 3, 2026, 6 states have implemented new maps, 4 have introduced legislation, and 4 are waiting on court orders.
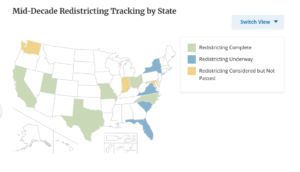
What is Virginia Trying to Do?
The proposed constitutional amendment would give the Virginia General Assembly the temporary authority to redraw congressional districts if another state redraws its districts for reasons other than completing decennial redistricting or complying with a court order. Since actions in Texas and other states have already triggered this “if” scenario, the results of the April 21 vote would have immediate impact.
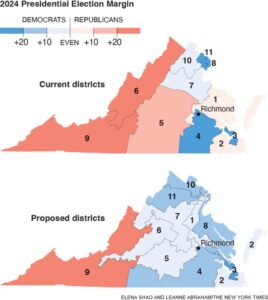
The Old Dominion with New Lines
Democratic leaders proposed a new map that passed the General Assembly and approved by Governor Abigail Spanberger. That map would reduce the current 5 majority-Republican districts to 1, meaning Representatives Rob Whittman, Jen Kiggans, John McGuire, and Ben Cline would be in danger of not being reelected. Rep. Morgan Griffith’s district would essentially remain the same.
Virginia Isn’t Feeling Like It’s for Lovers Right Now
Virginians can’t drive, watch TV, or pick-up their mail without being reminded of this high-stakes vote. Democrats claim the amendment is a justified response to Republican interference with congressional maps in other states. Former President Barack Obama has appeared in ads advocating for the new map, saying he believes this could help level the playing field between Republicans and Democrats.
On the contrary, Republicans condemn how the change will decrease Republican representation in Congress and could undermine the power of rural votes. The proposed lines expand the current rural districts to include urban centers that tend to vote blue, which would over-power the voice of red-leaning rural voters.
Does Money Talk?
So far, the Democratic-led Virginia for Fair Elections has out-raised the Republican-led Virginia for Fair Maps $64 million to $20 million. The vote is expected to be close, but Democrats currently hold the edge according to polling from the Washington Post and George Mason University which found 53 percent of registered VA voters plan to vote for the amendment and 44 percent said they plan to vote against the amendment.
What Are the Implications for 2026?
With a razor-thin majority in the US House, Virginia has clear and direct implications for the balance of power.
Specific to health care, Rep. Kiggans currently serves on the VA Health Subcommittee, Rep. McGuire is a member of the Oversight Health Care Subcommittee, and Rep. Cline is on the Appropriations subcommittee with a focus on the FDA. Rep. Griffith’s position as the lead Republican on the House Energy and Commerce Health subcommittee would likely not be in jeopardy.
Looking Ahead
Without a Republican House, President Trump will likely spend the last two years of his term fighting subpoenas and impeachment votes. A more partisan 2027 and 2028 (is that even possible?) could impact the mood of voters in the 2028 election. Although President Trump will no longer be on the ballot, his popularity and legacy will weigh on the minds of Republican primary voters and the general election voters.
On April 9 and 10, 2026, the Medicaid and CHIP Payment and Access Commission (MACPAC) met for their April meeting. The Commissioners held sessions on implementing community engagement requirements in Medicaid and on Medicaid program integrity. Commissioners also voted in favor of 2 recommendations related to Medicaid managed care to be included in the June 2026 report to Congress.
IMPLEMENTING COMMUNITY ENGAGEMENT REQUIREMENTS IN MEDICAID
MACPAC staff began the session by providing an overview of the previous research completed on the community engagement requirements, including expert panel and stakeholder interviews that were conducted in the summer of 2025. Staff then presented the draft recommendation that will be voted on during the May 2026 meeting, for inclusion in the June 2026 report to Congress. The draft recommendation reads: “The Secretary of the US Department of Health and Human Services should direct the Centers for Medicare & Medicaid Services (CMS) to develop a transparent plan for monitoring and evaluating community engagement requirements in Medicaid that provides insight into how such policies affect eligibility and enrollment, health status, employment, state and federal administrative spending, and the attainment of other identified policy goals. CMS should identify new metrics for state reporting, as needed, and build upon existing data collection activities to minimize administrative burden. Additionally, CMS should ensure timely publication of monitoring and evaluation results to inform policy and operational decision making.”
Commissioners appreciated the content and tone of the draft chapter, noting that the topic requires considerable detail. In general, the Commissioners indicated support for the draft recommendation but had some concerns about implementation. For example, Commissioners raised concerns about the additional administrative burden that could be placed on states due to monitoring and evaluation requirements. Commissioners commented that state Medicaid agencies lack the ability to track health insurance enrollment and health status once a beneficiary leaves the program. Commissioners did note that it is easier to track the health and insurance status of beneficiaries who continually enter and exit the program because data such as their use of preventive care, emergency department care, and health insurance coverage is often available from the time they were not enrolled in Medicaid. Overall, Commissioners felt it was important for the recommendation to be realistic about what data can be captured and not recommend beyond what is possible.
Many Commissioners also felt it would be important to evaluate the cost-effectiveness of community engagement requirement implementation, specifically understanding how much states are spending versus saving. Commissioners highlighted costs associated with the accelerated implementation timeline and the likely increase in uncompensated care, especially in rural communities that serve a higher percentage of Medicaid beneficiaries.
Commissioners also discussed a few other topics. One Commissioner highlighted the need to track the reason an individual becomes disenrolled after the work requirements are implemented, especially to understand if it is an administrative error and the individual is still eligible. Another Commissioner questioned whether there were platforms that could evaluate participation in nontraditional employment, such as gig work, as Equifax, one of the highlighted platforms, does not have that capability. MACPAC staff shared that beneficiaries could use an income verification service but that they are not currently aware of additional platforms to recommend. Commissioners and MACPAC staff agreed that new platforms may have become available since data was collected, and so additional research would be beneficial. Lastly, a Commissioner requested that staff include more literature on the effects of increased administrative burden on patients, specifically how it leads to higher rates of health insurance disenrollment. MACPAC staff agreed that including literature about the topic could be beneficial and said they would review possible inclusions.
INTRODUCTION TO MEDICAID PROGRAM INTEGRITY
This session began with MACPAC staff providing an overview of program integrity and fraud, waste, and abuse within Medicaid. Staff then discussed the roles and responsibility of federal and state departments in program integrity and outlined the current issues in Medicaid program integrity. Key takeaways from the presentation included:
-
Known fraud, waste, and abuse accounts for a small portion of program spending and its scale and impact are difficult to measure.
-
Federal government, states, and health plans have a significant number of statutory and regulatory program integrity responsibilities.
-
Key issues in program integrity include federal-state collaboration and program integrity in managed care.
Staff requested that the Commissioners provide feedback on the policy scan findings and further areas for exploration during stakeholder interviews.
Commissioners widely praised the material presented and appreciated the clear definitions and examples of fraud, waste, and abuse. Multiple Commissioners expressed the view that often the terms are inaccurately used, pointing out that disagreeing with a policy or statute does not constitute fraudulent or wasteful spending.
Commissioners had a wide variety of topics of interest for further exploration. A few Commissioners were interested in understanding how CMS and states decide where to invest money and time to best prevent program integrity issues and recover improper payments. One Commissioner specifically requested a return on investment analysis to understand where to invest to produce the best results. There was also widespread interest in understanding best practices for preventing fraud, recovering payments, and working with state Medicaid agencies.
Some other areas of interest included measuring the quality of State Medicaid Fraud Control Units, understanding how prior authorization is useful for program integrity, the impact of delays in convictions during fraud investigations for states, and ways to improve collaboration between CMS and states.
VOTE ON RECOMMENDATIONS FOR JUNE REPORT TO CONGRESS
Commissioners voted on 2 recommendations related to ensuring accountability of Medicaid managed care plans for inclusion in the June 2026 report to Congress.
The first recommendation reads, “The Secretary of the US Department of Health and Human Services should direct the Centers for Medicare and Medicaid Services to provide guidance on the types of accountability actions, such as liquidated damages, informal interventions, and other accountability actions, taken in response to plan noncompliance, in the sanctions section of the Managed Care Program Annual Report pursuant to 42 CFR 438.66(e)(2)(viii).” This recommendation passed by a vote of 17 to 0.
The second recommendation reads, “The Secretary of the US Department of Health and Human Services should direct the Centers for Medicare and Medicaid Services to develop a publicly available database on managed care plan performance that links federally mandated reported data together to facilitate analysis. CMS should also issue guidance and toolkits to help states effectively use these data to assess past performance, improve beneficiary experience, and oversee managed care plans.” This recommendation passed by a vote of 17 to 0.
There was no discussion about either of the recommendations.
On April 10, 2026, the Centers for Medicare and Medicaid Services (CMS) released the Fiscal Year 2027 Medicare Hospital Inpatient Prospective Payment System (IPPS) Proposed Rule. A fact sheet from CMS is available here. Complete text of the proposal is available here. Comments on the rule are due June 9, 2026.
UPDATES TO IPPS PAYMENT RATES
As part of the proposed rule, CMS would increase payment rates by 2.4% for general acute care hospitals that successfully participate in the Hospital Inpatient Quality Reporting (IQR) Program and are meaningful users of electronic health records (EHRs) under the Medicare Promoting Interoperability Program. This proposed update reflects a projected FY 2027 hospital market basket increase of 3.2%, reduced by a 0.8% productivity adjustment. Overall, CMS estimates that the proposed changes in IPPS payment rates, together with other policy changes, would increase hospital payments by approximately $1.4 billion in FY 2027.
CMS also proposes setting the FY 2027 outlier fixed-loss threshold at $51,704, which would target outlier payments at approximately 5.14% of total operating DRG payments and capital payments, after incorporating an estimate of outlier reconciliation. In its impact analysis, CMS estimates that overall IPPS payments would increase by 1.2% relative to FY 2026, reflecting the combined effects of the proposed update, outlier policies, uncompensated care payments, and wage index changes.
MEDICARE-DEPENDENT HOSPITALS (MDHS) AND LOW-VOLUME HOSPITALS
The MDH program provides enhanced payments to small rural hospitals (≤100 beds, not Sole Community Hospitals) where at least 60% of inpatient days or discharges are attributable to Medicare patients, using a blended rate that includes 75% of the federal rate plus 25% of the hospital-specific rate based on historical costs. The low-volume hospital adjustment offers percentage add-ons to IPPS payments for rural hospitals with low annual discharges to offset higher per-case costs, currently on a sliding scale up to 25% for facilities with fewer than 3,800 discharges and more than 15 road miles from another subsection (d) hospital.
In the FY 2027 proposed rule, CMS does not propose substantive policy changes to either the MDH program or the low-volume hospital adjustment. Rather, the agency proposes conforming regulatory changes to reflect current law, under which both policies have been extended through December 31, 2026. As a result, absent further congressional action, the MDH program would expire, and the temporary low-volume hospital policy would revert to permanent law beginning January 1, 2027.
Accordingly, the central issue is less the substance of CMS’s proposal than the current appropriations and extender status of these programs. Congress first provided a short-term extension in the Continuing Appropriations, Agriculture, Legislative Branch, Military Construction and Veterans Affairs, and Extensions Act, 2026, and subsequently extended both policies through December 31, 2026, in the Consolidated Appropriations Act, 2026. CMS states that it will revise the regulatory language in the final rule if Congress again extends the programs before the rule is finalized. In practical terms, unless lawmakers enact another extender later this year, both policies will lapse beginning January 1, 2027.
CMS estimates that, if Congress were to extend the MDH and low-volume hospital policies through the end of FY 2027, affected hospitals would receive approximately $0.4 billion in additional payments. Conversely, under current law, CMS estimates that expiration of the MDH status would reduce payments by approximately $110 million for roughly 80 affected hospitals, while expiration of the temporary low-volume policy would reduce aggregate payments by approximately $258 million in FY 2027, with approximately 589 hospitals expected to lose qualification under the stricter post-January 1 criteria.
DISCONTINUATION OF THE LOW-WAGE INDEX HOSPITAL POLICY
The low-wage index hospital policy was established in the FY2020 IPPS final rule as a temporary, budget-neutral initiative to address wage index disparities, benefiting rural hospitals by raising their wage indices to mitigate the impacts of lower payments. This policy adjusted the wage index for hospitals in the bottom quartile, setting a floor at the 25th percentile value, which was offset by a corresponding reduction for higher-wage hospitals. However, in July 2024, the U.S. Court of Appeals for the D.C. Circuit in Bridgeport Hosp. v. Becerra ruled that CMS lacked the statutory authority under sections 1886(d)(3)(E) or 1886(d)(5)(I) of the Social Security Act (SSA) to implement this policy, vacating both the policy and its budget neutrality adjustment.
In the FY 2026 final rule, CMS formally discontinued the low-wage index hospital policy and its associated budget-neutrality adjustment for FY 2026 and subsequent fiscal years. At the same time, CMS finalized a narrow, budget-neutral transitional payment exception for FY 2026 for certain hospitals experiencing significant decreases in their wage index resulting from the policy’s discontinuation. Under that transition, eligible hospitals could receive additional FY 2026 payments if their wage index otherwise would have fallen by more than 9.75% from their FY 2024 wage index, with payments calculated as if the hospital’s FY 2026 wage index were equal to 90.25% of its FY 2024 wage index.
For FY 2027, CMS does not propose to reinstate the low-wage index hospital policy. Instead, it proposes continuing the same transition approach for hospitals that are still significantly affected by the policy’s discontinuation. Specifically, CMS proposes a narrow transitional payment exception for hospitals whose FY 2027 wage index would be more than 14.2625% below their FY 2024 wage index, with those hospitals receiving FY 2027 payments as if their wage index were equal to 85.7375% of their FY 2024 wage index. CMS proposes implementing this transition in a budget-neutral manner after applying the 5% cap on wage index decreases.
HOSPITAL INPATIENT QUALITY REPORTING (IQR) PROGRAM
In the FY 2027 proposed rule, CMS proposes a fairly broad set of updates to the Hospital IQR Program. Most notably, CMS would adopt three new measures: the Excess Days in Acute Care After Hospitalization for Diabetes measure beginning with the FY 2029 payment determination, and the Hospital Harm–Postoperative Venous Thromboembolism eCQM and Advance Care Planning eCQM beginning with the FY 2030 payment determination. CMS also proposes to adopt modified versions of five mortality measures beginning with the FY 2028 payment determination – acute myocardial infarction, heart failure, pneumonia, COPD, and CABG mortality – by adding Medicare Advantage patients and shortening the applicable performance period from three years to two years. In addition, CMS proposes similar modifications to three Excess Days in Acute Care measures (for AMI, heart failure, and pneumonia), also beginning with the FY 2028 payment determination.
CMS also proposes to remove three eCQMs beginning with the FY 2030 payment determination: VTE-1, VTE-2, and STK-02. On reporting requirements, CMS proposes to make reporting of the Malnutrition Care Score eCQM mandatory beginning with the FY 2030 payment determination and to establish a policy under which Hospital Harm eCQMs would become mandatory after two years of self-selected reporting. CMS also proposes to update the Maternal Morbidity Structural Measure, beginning with the FY 2028 payment determination, so that hospitals identify which perinatal quality collaborative program they participate in. The proposed rule explains that these reporting changes are intended to continue CMS’s move toward a more digital measure set and to standardize hospital quality reporting in several areas.
MEDICARE PROMOTING INTEROPERABILITY PROGRAM
In the FY 2027 proposed rule, CMS proposes a series of changes to the Medicare Promoting Interoperability Program, largely focused on streamlining the program’s certification and attestation requirements and shifting hospitals toward newer interoperability functions.
-
Most notably, CMS would update the definition of certified electronic health record technology (CEHRT) to align with changes proposed by Office of the National Coordinator for Health Information Technology (ONC), including removing references to several certification criteria from the program’s CEHRT definition.
-
CMS also proposes to remove the ONC Direct Review and ONC-Authorized Certification Body surveillance attestations beginning with the CY 2026 EHR reporting period, which the agency describes as a burden-reduction step.
-
In addition, CMS proposes to remove the Support Electronic Referral Loops by Sending Health Information and Support Electronic Referral Loops by Receiving and Reconciling Health Information measures beginning with the CY 2028 EHR reporting period, on the view that newer network-based exchange measures better reflect meaningful health information exchange
CMS also proposes several more targeted policy updates.
-
The agency would modify the Electronic Prior Authorization measure, including revising the measure language to require that prior authorization be requested electronically using CEHRT and changing the reference from “discharge” to “encounter.”
-
CMS further proposes to make that measure optional and worth 10 bonus points for CY 2027, before requiring hospitals to attest “Yes” beginning with the CY 2028 EHR reporting period. CMS indicates the measure would remain unscored in CY 2028 and subsequent years for purposes of point allocation.
-
Separately, CMS proposes to add a new Unique Device Identifiers for Implantable Medical Devices measure under the Public Health and Clinical Data Exchange objective beginning with the CY 2027 EHR reporting period.
-
Finally, in alignment with the Hospital IQR Program, CMS proposes to adopt two new eCQMs (the Hospital Harm-Postoperative Venous Thromboembolism eCQM and the Advance Care Planning eCQM) beginning with the FY 2030 payment determination, and to remove three eCQMs (VTE-1, VTE-2, and STK-02), also beginning with the FY 2030 payment determination.
HOSPITAL READMISSIONS REDUCTION PROGRAM
In the FY 2027 proposed rule, CMS proposes one substantive change to the Hospital Readmissions Reduction Program (HRRP): adoption of the Hospital 30-Day, All-Cause, Risk-Standardized Readmission Rate Following Sepsis Hospitalization measure. CMS would provide hospitals with an early look at performance for the FY 2028 program year and then begin using the measure for payment adjustment purposes in FY 2029. CMS states that it proposes moving the measure directly into HRRP, rather than phasing it in first through a reporting-only program, because sepsis readmissions are associated with significant morbidity. Under the proposal, the FY 2028 early look would be based on an applicable period of July 1, 2024, through June 30, 2026. The measure would first be used for FY 2029 payment adjustments.
HOSPITAL VALUE-BASED PURCHASING (VBP) PROGRAM
In the FY 2027 proposed rule, CMS does not propose changes to the current Hospital Value-Based Purchasing (VBP) measure set for the FY 2027 program year. CMS instead proposes substantive updates to five existing mortality measures in the Clinical Outcomes domain, beginning with the FY 2032 program year: the Hospital 30-Day, All-Cause, Risk-Standardized Mortality Rate Following Acute Myocardial Infarction (AMI) Hospitalization, Heart Failure Hospitalization, Pneumonia Hospitalization, Chronic Obstructive Pulmonary Disease (COPD) Hospitalization, and Coronary Artery Bypass Graft (CABG) Surgery measures.
For these five measures, CMS proposes adding MA beneficiaries to the measure population and shortening the performance period from three years to two years. The proposed rule also includes a related technical update to the risk adjustment methodology for these measures, replacing the current use of hierarchical condition categories with individual ICD-10 codes. CMS states that these VBP changes would be contingent on first adopting the same refined measures in the Hospital Inpatient Quality Reporting (IQR) Program, consistent with the statutory requirement that measures be publicly reported before being used in the VBP Program.
REQUESTS FOR INFORMATION (RFIS)
CMS includes several RFIs aimed at shaping potential future quality program and model design decisions.
-
Measuring Emergency Care Access and Timeliness in the Hospital Inpatient Quality Reporting and Hospital Value-Based Purchasing Programs: CMS seeks feedback on whether and how the Emergency Care Access & Timeliness eCQM could be used in the inpatient quality programs, including barriers to improving bed availability and ED boarding, whether the outpatient-developed measure specifications fit the inpatient setting, and whether any numerator, denominator, exclusion, or accountability changes would be needed before future adoption.
-
Potential Future Use of the Adult Community-Onset Sepsis Standardized Mortality Ratio Measure in the Hospital Inpatient Quality Reporting Program: CMS seeks comment on the possible future use of this sepsis mortality measure in IQR, including operational feasibility, workflow and data challenges, how claims and EHR/FHIR data would be reconciled, and what implementation issues hospitals (particularly rural hospitals) might face.
-
Birthing-Friendly Hospital Designation Modification to Expand Designation Criteria: CMS seeks feedback on expanding the Birthing-Friendly Hospital designation to incorporate the Cesarean Birth eCQM and Severe Obstetric Complications eCQM, as well as on a revised scoring methodology, peer grouping, tiered designation icons, and how the designation should be displayed for consumers.
-
Ambulatory Surgical Center (ASC) Episode Request for Information: In the TEAM section, CMS seeks comment on whether and how ASCs could be incorporated into the model in future years, including questions about model structure, participant roles, financial accountability, episode design, target pricing, quality measurement, and whether adding ASCs would require a separate model test.
-
Hospital with Physician Ownership Request for Information: Also in the TEAM section, CMS seeks feedback on whether physician-owned hospitals should be allowed to opt into TEAM voluntarily in future years, and, if so, what eligibility criteria, guardrails, and participation requirements should apply to protect model integrity and Medicare savings.

